With over 10 years of experience supporting 100+ clinical trials and collaborating with 50+ sponsors and research sites, OnePath Research Services delivers audit-ready documentation, streamlined workflows, and compassionate participant resources — all designed to advance your clinical research journey.
Expert support for smooth, compliant trial setups.
Help maintain audit-ready documentation.
Helping you feel informed, prepared, and supported.
OnePath Research Services is a specialized consulting firm providing documentation, compliance, and operations support to organizations involved in clinical research and regulated industries. We help CROs, sponsors, and small research-focused businesses streamline their workflows, strengthen audit readiness, and maintain regulatory alignment through high-quality documentation and support tools.
Our services include SOP creation, site and study-level trackers, slide deck formatting, regulatory document reviews, and document automation strategies. We work with precision and discretion—delivering polished, audit-ready materials designed to support operational excellence.
In addition to our professional services, OnePath proudly offers free support services for clinical trial participants and patients.
We help individuals understand consent forms, identify relevant clinical trials, and serve as a compassionate resource—offering guidance, clarity, and emotional support throughout their research journey.


We help sponsors, CROs, and clinical trial sites achieve inspection-ready documentation through structured regulatory support. From TMF audits to SOP development and site startup readiness, our approach ensures compliance, clarity, and operational efficiency at every stage of the study.
Aligned with CDISC TMF & ISF standards and evolving ICH E6(R3) regulatory expectations.
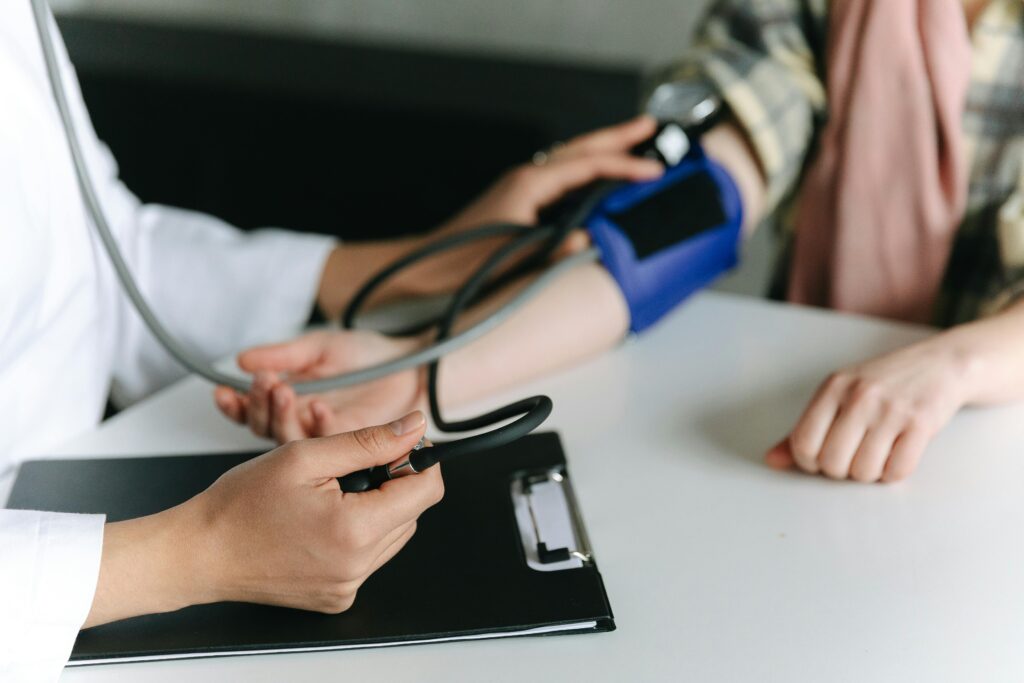
We provide tools and resources to help patients understand clinical trials and feel supported every step of the way. From preparing for informed consent to navigating transportation or follow-up visits, we make participation more accessible, inclusive, and compassionate.

Our ready-to-use regulatory templates are designed for clinical researchers, coordinators, and startup sites looking to streamline operations. From CV formats and DOA logs to TMF QC checklists and SOPs, each tool is professionally formatted and trial-site ready.

We prioritize detail, clarity, and compliance in every deliverable.
Our service reflects the high standards of the industry we support.
We operate with discretion and care, protecting sensitive data and proprietary information.
Clients count on us for consistent, timely, and high-quality execution.
We view our role as a partner, not just a provider—here to help research move forward.
We are committed to supporting the real people behind every trial, treating patients and participants with empathy and respect.


OnePath Research Services combines deep regulatory expertise with structured workflows and AI-assisted tools to improve accuracy, streamline documentation, and support efficient clinical trial execution.
We help sponsors, CROs, and trial sites operate more effectively by enhancing submission readiness, strengthening document quality, and reducing operational gaps.
Feedback based on professional experience supporting sponsors, study teams, and clinical research operations.
Trusted by sponsors, CROs, and clinical research sites to deliver reliable, high-quality operational support.
Highly supportive and easy to work with. Provided clear guidance throughout the process, making complex tasks manageable for site teams.
Consistently professional, organized, and proactive. Communication was clear, concise, and action-oriented, making it easy for teams and sites to stay aligned.
Efficiency, organization, and autonomy made a significant impact on our team. Consistently delivered high-quality work while maintaining flexibility and strong collaboration.
Went above and beyond to ensure critical timelines were met. Even in urgent situations, everything was handled with professionalism, speed, and precision.
A dependable and proactive team member who consistently delivers. Ability to stay calm under pressure and keep everything moving forward made a noticeable difference.
Demonstrated exceptional ability to manage high-priority situations under pressure. Turnarounds were fast, solutions were effective, and expectations were consistently exceeded.
Testimonials reflect professional experience across prior clinical research engagements.